Rat Carotid Artery Endothelial Cells
Cat.No.: CSC-C8698W
Species: Rat
Source: Artery
Cell Type: Endothelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat carotid artery endothelial cells (RCAECs) were cultured from normal carotid artery tissue in laboratory rats. The carotid artery, the blood vessel in the neck of vertebrates, divides into the external carotid artery and the internal carotid artery. External carotid artery is for the head and face, and internal carotid artery is through the skull to the brain and eyes. These cells cover the inside of the carotid artery and provide a natural barrier between blood and other tissues. RCAECs are endothelial, and either polygonal or spindle-shaped. They are monolayers on culture plates and are bound by narrow and tight junctions.
RCAECs might be similar to human carotid endothelial cells in some way, including being sensitive to hemodynamic fluctuations. For instance, studies show that under blood flow induction, rat carotid endothelial cells undergo reprogramming, exhibiting heterogeneity and dynamism. Rat carotid models are widely used in atherosclerosis and cardiovascular disease research. Studies also indicate that these cells demonstrate significant biological properties in inflammation, oxidative stress, and apoptosis.
Static Targeting of S1P@CD-PLGA NBs to Inflammatory Rat Carotid Endothelial Cells
Vascular inflammation, a common pathophysiological element in life-threatening cardiovascular and cerebrovascular conditions, demands precise targeting for effective diagnosis and treatment. Given the high expression of S1P receptors in pathological states, Tang's team deployed polylactic acid-co-glycolic acid (PLGA) nanobubbles encapsulating SF6 and S1P. The design capitalizes on S1P's natural high-affinity interactions with S1PRs for targeted delivery to inflammatory vascular endothelial cells.
The characterization results showed that S1P@CD-PLGA NBs with diameters of ≈200 nm have good stability, biosafety, and ultrasound imaging-enhancement effects. Then, rat carotid artery endothelial cells were used to evaluate S1P@CD-PLGA NBs, with histamine inducing inflammation. Figure 1a confirms the NBs' biocompatibility and noncytotoxicity, showing no adverse effects on cell viability at concentrations up to 0.60 mg/mL over various times. Therefore, 0.60 mg/mL was chosen for further experiments. DiI-labeled S1P@CD-PLGA NBs showed rapid targeting of inflammatory endothelial cells with notable fluorescence intensity differences at various incubation times (Fig.1b). This indicates a specific targeting effect and time-dependent uptake, higher in inflammatory conditions due to increased vascular permeability. Laser confocal microscopy (Fig. 1c) and TEM (Fig. 1d) confirmed membrane targeting and intracellular uptake, showing enhanced accumulation in inflammatory cells compared to CD-PLGA NBs under identical conditions.
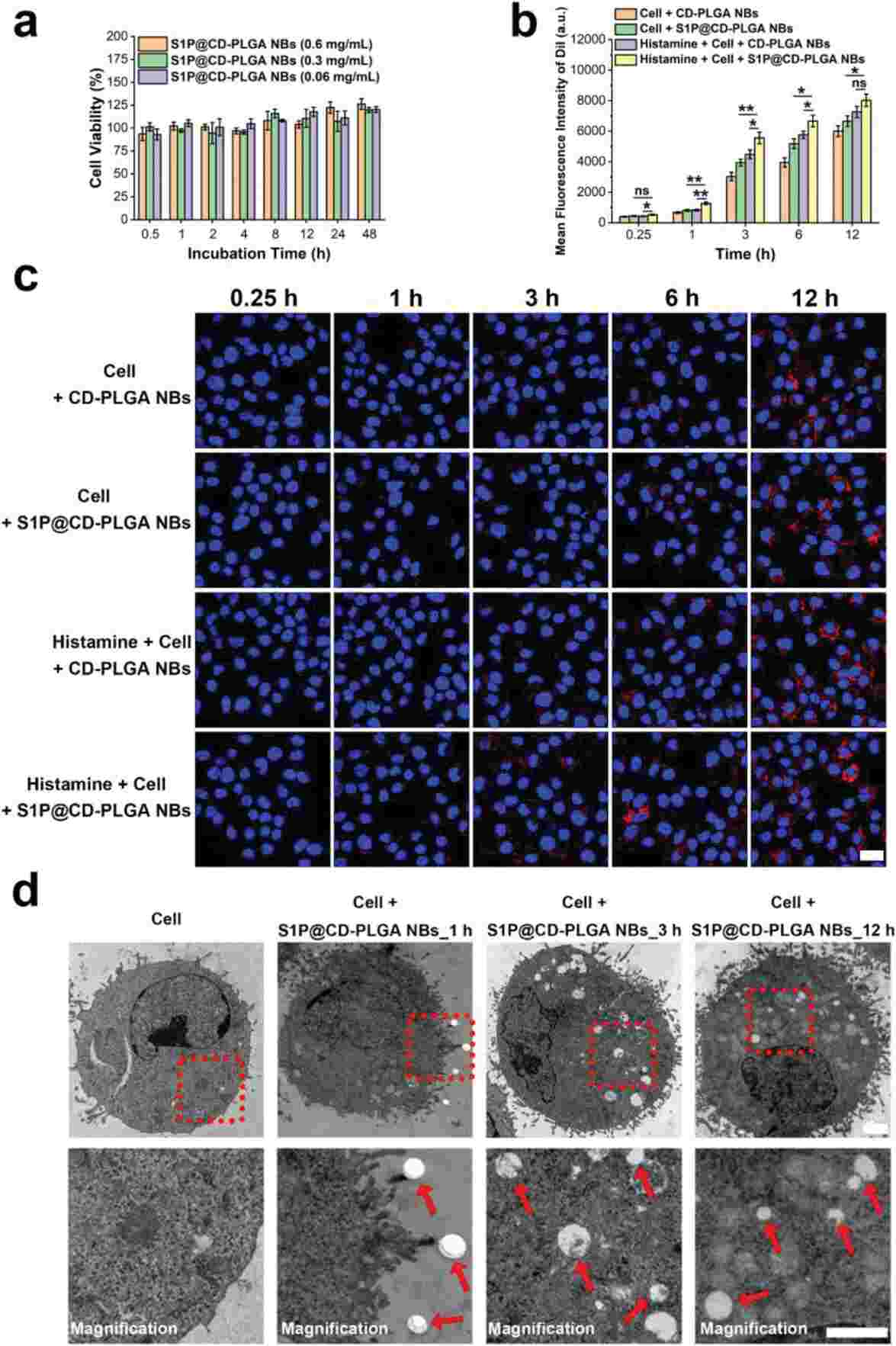 Fig. 1. The toxicity and static targeting of S1P@CD-PLGA NBs to rat carotid endothelial cells (Tang J, Liu Y, et al., 2023).
Fig. 1. The toxicity and static targeting of S1P@CD-PLGA NBs to rat carotid endothelial cells (Tang J, Liu Y, et al., 2023).
Biosafety Evaluation of S1P@CD-PLGA-Rtpa Nbs on Rat Carotid Endothelial Cells
Thrombotic diseases pose serious health risks, yet current treatments remain suboptimal due to drug targeting and stability issues. Tang's team designed the S1P@CD-PLGA-rtPA NBs, a nanocarrier integrating SF6-loaded PLGA NBs, cyclodextrin, S1P, and rtPA.
They tested biosafety of the S1P@CD-PLGA-rtPA NBs on rat carotid endothelial cells. First, the toxicity of S1P@CD-PLGA-rtPA NBs was assessed at various concentrations (0.2, 1, and 5 mg/ml). Fig. 2D shows no adverse effects on cell viability over time, confirming their biocompatibility and non-toxicity. Second, the hemolysis effect of these NBs on erythrocytes was evaluated. As seen in Fig. 2E, hemolysis rates were 1.67% (0.2 mg/ml), 2.22% (1 mg/ml), and 2.78% (5 mg/ml), all under 5%, indicating no significant hemolysis. Third, platelet aggregation tests indicated that while thrombin caused rapid aggregation, both saline and NBs did not (Fig. 2F), suggesting a reduced risk of secondary thrombosis. These findings confirm the excellent hemocompatibility of S1P@CD-PLGA-rtPA NBs for in vivo thrombolytic applications.
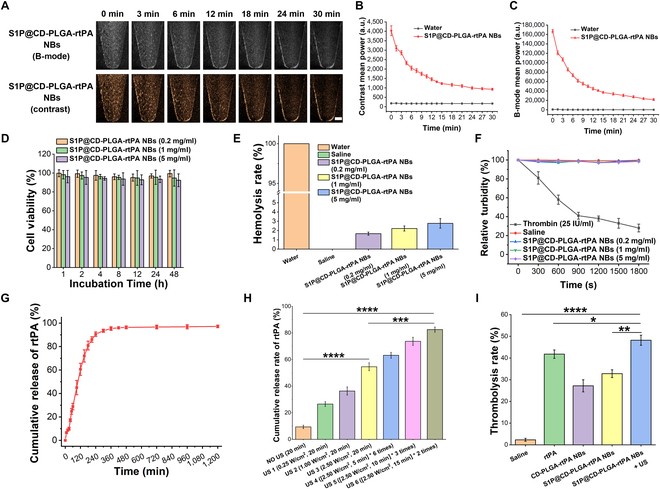 Fig. 2. Characterization of in vitro ultrasound imaging enhancement, biosafety, in vitro release of rtPA, and in vitro thrombolysis of S1P@CD-PLGA-rtPA NBs (Tang J, Xu H, et al., 2024).
Fig. 2. Characterization of in vitro ultrasound imaging enhancement, biosafety, in vitro release of rtPA, and in vitro thrombolysis of S1P@CD-PLGA-rtPA NBs (Tang J, Xu H, et al., 2024).
Ask a Question
Write your own review