ONLINE INQUIRY
Rat Bone Marrow-Derived Endothelial Cells
Cat.No.: CSC-C8697W
Species: Rat
Source: Bone Marrow
Cell Type: Endothelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat bone marrow-derived endothelial cells (BM-ECs) are typically isolated from the femur or tibia of the rats. Bone marrow, a soft tissue that lies in the bony cavity, is primarily a hematopoietic cell source – containing stem and progenitor cells as well as precursor cells that can develop into endothelial cells. These precursor cells, endothelial progenitor cells (EPCs) or angioblasts, are migratory immature endothelial cells that can continue to grow and differentiate. BM-ECs can, given the right conditions, grow into endothelial cells, carrying markers such as CD133, CD146 and VEGFR-2, and develop cell layers that resemble normal endothelial cells. Moreover, in certain cultures BM-ECs grow into the shape of capillaries and have high angiogenic activity in vascular growth assays.
During tumor angiogenesis, BM-ECs are recruited to tumor sites, where they differentiate into mature endothelial cells involved in new blood vessel formation. Studies suggest that they play a critical role in angiogenesis in early tumors, but can be replaced by other types of endothelial cells in late tumors. Furthermore, in metabolic syndrome models, the migration potential of BM-ECs is influenced by insulin resistance, suggesting that they might be involved in the pathogenesis of diabetes-associated vascular lesions.
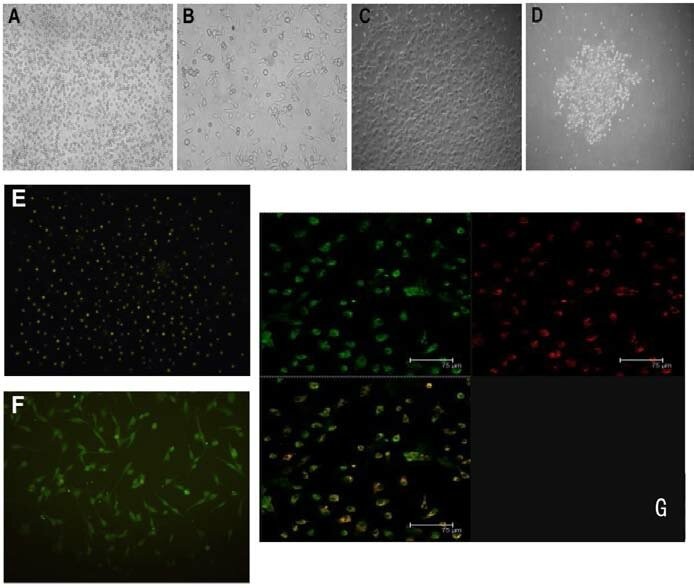 Fig. 1. Rat bone marrow-derived endothelial progenitor cells culture, expansion and identification. After 2-3 weeks, cell colonies gradually formed (Figs. 1A-D). (A: 1st day; B: 5th day; C: 14th day; D: 20th day); under fluorescent microscopy, CD34+ cells (Fig. 1E) and vWF+ cells (Fig. 1F) fluoresced green; under confocal microscopy, red (Dil-Ac-LDL) and green (FITC-UEA-lectin-1) dual-fluorescent-positive cells were considered to be EPCs (Fig. 1G) (Wang Y, Song J, et al., 2014).
Fig. 1. Rat bone marrow-derived endothelial progenitor cells culture, expansion and identification. After 2-3 weeks, cell colonies gradually formed (Figs. 1A-D). (A: 1st day; B: 5th day; C: 14th day; D: 20th day); under fluorescent microscopy, CD34+ cells (Fig. 1E) and vWF+ cells (Fig. 1F) fluoresced green; under confocal microscopy, red (Dil-Ac-LDL) and green (FITC-UEA-lectin-1) dual-fluorescent-positive cells were considered to be EPCs (Fig. 1G) (Wang Y, Song J, et al., 2014).
MTX Treatment Alters Notch Signaling in Cultured Rat BMECs and Blocking Notch2 Attenuates MTX-Induced Notch Target Gene Hey1 Overexpression
Chemotherapy, particularly with methotrexate (MTX), is known to lead to bone vasculature dysfunction and associated complications like osteoporosis and increased fracture risks. These vascular damages stem from compromised endothelial cells and are exacerbated by MTX. Paymanfar's team examined the Notch2 signaling pathway's involvement in bone micro-vasculature impairment caused by MTX, utilizing a rat model and rat bone marrow endothelial cell (BMEC) models.
The results on the rat model are shown that MTX-induced micro-vasculature dilation and regression is associated with the induction of Notch2 activity in endothelial cells and increased production of inflammatory cytokine tumor necrosis factor alpha (TNFα) from osteoblasts (bone forming cells) and bone marrow cells. Blockade of Notch2 by a neutralizing antibody ameliorated MTX adverse effects on bone micro-vasculature, both directly by supressing Notch2 signaling in endothelial cells and indirectly via reducing TNFα production. Then, using rat bone marrow endothelial cell (BMEC) models, they confirmed Notch2 signaling role in MTX-induced bone vasculature damage. Cultured BMECs were treated with saline or MTX (10 µM) for 24 hours, followed by RT-PCR analysis of Notch ligand Jag1 and target gene Hey1. Results showed significant increases in Jag1 (Fig. 1A) and Notch2 (Fig. 1B) mRNA in MTX-treated BMECs (p < 0.05). Hey1 mRNA was also significantly upregulated (p < 0.01) (Fig. 1C). To assess if MTX-induced Hey1 mRNA increase was linked to Notch2, and whether anti-Notch2 antibody could suppress Hey1 expression, treatments were repeated. Hey1 mRNA upregulation was confirmed and reduced with Notch2 blockade (p < 0.001 vs. MTX+Control IgG) (Fig. 1D), indicating Notch2 blockade inhibits MTX-induced Hey1 increase.
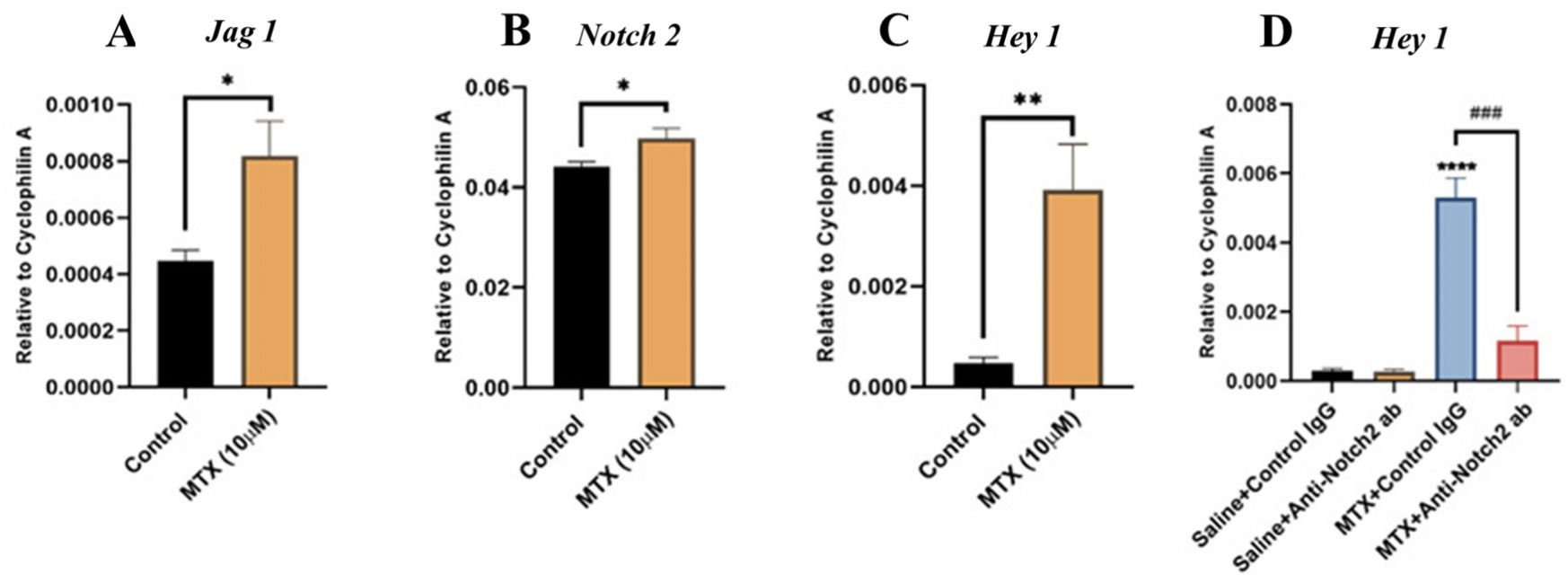 Fig. 1. MTX treatment effects on the Notch2 ligand Jag1, Notch2, and the Notch target gene Hey1 mRNA expression and the role of Notch2 signaling in MTX-induced Hey1 induction in cultured rat bone marrow-derived endothelial cells (BMECs) (Peymanfar Y, Su Y, et al., 2022).
Fig. 1. MTX treatment effects on the Notch2 ligand Jag1, Notch2, and the Notch target gene Hey1 mRNA expression and the role of Notch2 signaling in MTX-induced Hey1 induction in cultured rat bone marrow-derived endothelial cells (BMECs) (Peymanfar Y, Su Y, et al., 2022).
SNs Increase Matrix Metalloproteinases Production and Activity by ECs
Sensory nervous system contributes to the repair of bone - in particular, to angiogenesis and osteogenesis. But it doesn't yet know exactly what effects it has on ECs.
Leroux's lab cultured sensory neurons with endothelial cells using microfluidic apparatus, and measured gene expression, MMP protein and activity. They described how ECs are affected by sensory neurons (SNs) and neuropeptides such as calcitonin gene-related peptide (CGRP) and substance P (SP). By day 7, rat bone marrow endothelial cell (BMEC) co-cultured with sensory neurons (SNs) expressed Angpt1 (1.7-fold), VegfA (2-fold) and Col4 (2.8-fold) more frequently than ECs alone (Fig. 2a). Pecam1 expression remained unchanged. Notably, Mmp2 expression significantly increased at day 4 (6-fold) and day 7 (22-fold) in co-cultures with SNs (Fig. 2b). Protein levels of MMP2/MMP9 also significantly rose, showing a 13-fold increase at day 4 and an 8-fold increase at day 7 with SNs (Fig. 2b), corroborating Mmp2 gene upregulation. Zymography indicated heightened enzymatic activity and various MMP isoforms in ECs co-cultured with SNs (Fig. 2b).
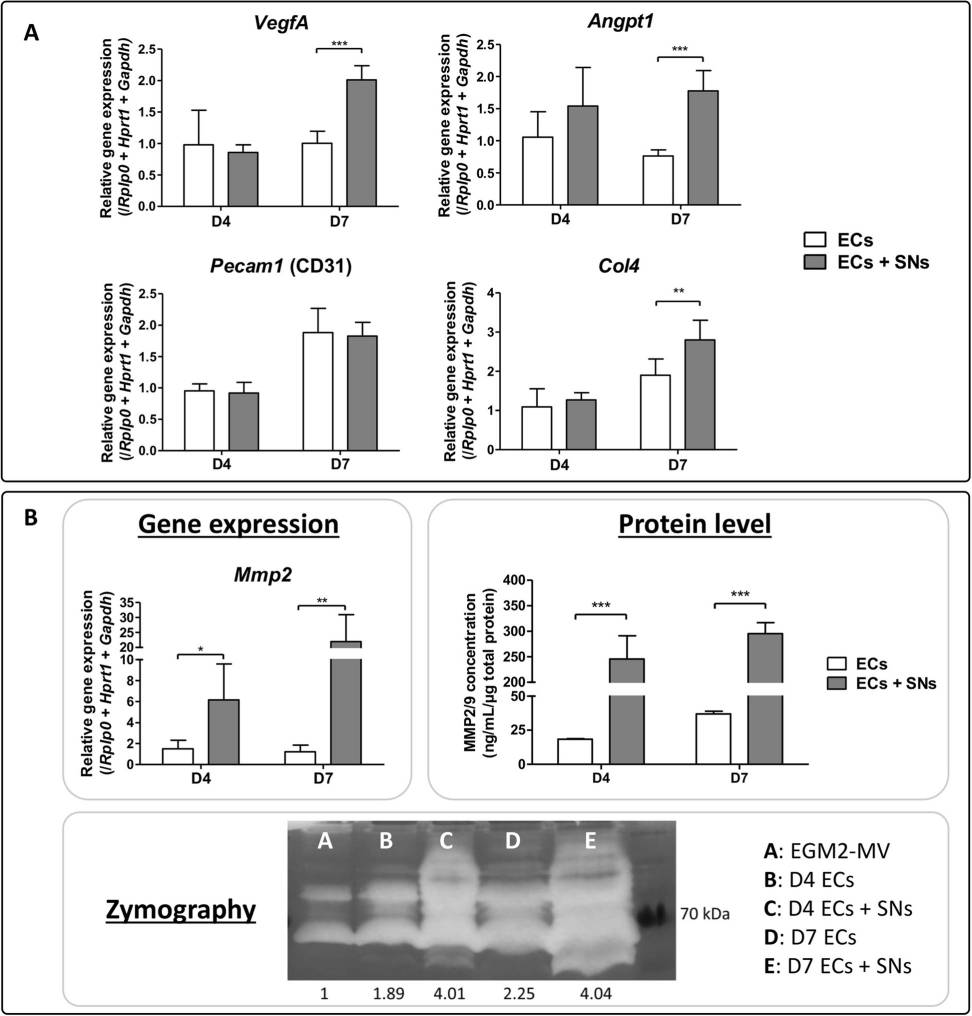 Fig. 2. Effect of CGRP and SP on ECs transcriptional profiles (Leroux A, Santos B P, et al., 2020).
Fig. 2. Effect of CGRP and SP on ECs transcriptional profiles (Leroux A, Santos B P, et al., 2020).
Ask a Question
Write your own review