Oral Mucositis Model
Creative Bioarray has dedicated decades to refining and perfecting a reliable oral mucositis model for our customers. Our meticulously developed model enables in-depth exploration of the mechanisms behind oral mucositis and facilitates the screening of promising new drug candidates. Leveraging our unparalleled platforms and extensive expertise, we are committed to delivering cost-effective and high-quality services to meet your research needs.
Radiotherapy, a widely practiced treatment for cancer, involves administering lethal doses of radiation specifically targeted at cancerous cells. Nevertheless, the exposure of healthy tissue to radiation can trigger both acute and chronic toxicities, manifesting as dermatitis, oral mucositis, altered taste sensation, pain, xerostomia, decreased appetite, and even ulceration. Oral mucositis stands out as one of the most prevalent complications arising from cancer therapy, chemotherapy, and radiotherapy. In patients suffering from granulocytopenia, this condition frequently leads to systemic infections and nutritional deficiencies due to dietary restrictions. Radiation-induced oral mucositis is triggered by a range of mechanisms, including but not limited to the release of free radicals, altered proteins, and proinflammatory cytokines such as interleukin-1β, prostaglandins, and tumor necrosis factor, which are secreted by irradiated epithelial, endothelial, and connective tissue cells within the buccal mucosa. Despite significant technological advancements, a successful approach to prevent radiation-induced oral mucositis and toxicity in normal cells remains elusive.
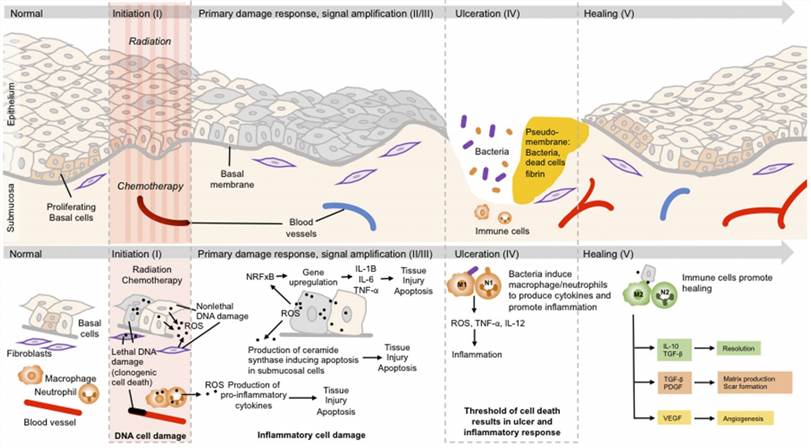 Fig. 1 Illustration of the 5 phases of mucositis
Fig. 1 Illustration of the 5 phases of mucositis
Our Oral Mucositis Model
- Available Animal
Rat
Golden hamster - Modeling Method
After anesthetization, the tongue of rats or the cheek pouch of golden hamsters receive single or repeated radiation exposure at doses ranging from 25-40 Gy. Other parts of the body are protected with a lead shield. - Endpoints
- Survival rate
- Food/Water intake
- Body weight
- Histology analysis: H&E staining, Toluidine blue staining, etc.
- Other customized endpoints: available upon request.
Example Data
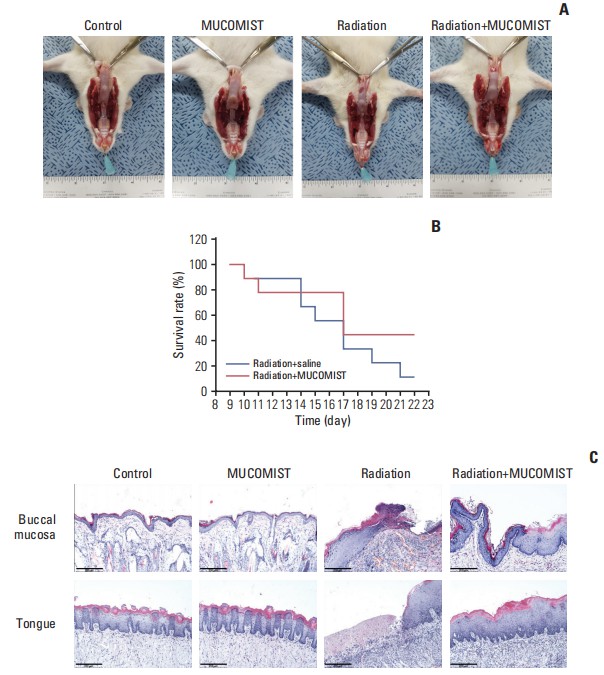 Fig. 2 Protective effect of N-acetyl cysteine (NAC) against histopathologic effects in irradiated rats. (A) Photographic images of buccal mucosa on day 23 after irradiation. Rats in the radiation group were found to have more tongue mucositis (the tip of the tongue is not fully healed). However, the MUCOMIST(NAC) treatment group is clean except for the tip of the tongue. (B) Survival rate and time of death. There was no statistical significance between the treatment and non-treatment groups. (C) Histopathologic images (H&E staining, ×400) of buccal mucosa and tongue on day 23 after irradiation.
Fig. 2 Protective effect of N-acetyl cysteine (NAC) against histopathologic effects in irradiated rats. (A) Photographic images of buccal mucosa on day 23 after irradiation. Rats in the radiation group were found to have more tongue mucositis (the tip of the tongue is not fully healed). However, the MUCOMIST(NAC) treatment group is clean except for the tip of the tongue. (B) Survival rate and time of death. There was no statistical significance between the treatment and non-treatment groups. (C) Histopathologic images (H&E staining, ×400) of buccal mucosa and tongue on day 23 after irradiation.
Quotation and Ordering
Creative Bioarray has an extensive range of established disease models that can be utilized to screen novel drug candidates. We are dedicated to helping our clients accelerate their development process. If you are interested in our services, please feel free to contact us at any time or submit an inquiry to us directly.
References
- Oronsky, B., et al. A Review of Clinical Radioprotection and Chemoprotection for Oral Mucositis. Transl Oncol. 2018, 11(3):771-778.
- Kim, H.J, et al. Protective effects of N-acetylcysteine against radiation-induced oral mucositis in vitro and in vivo. Cancer Research and Treatment: Official Journal of Korean Cancer Association, 2020, 52(4): 1019.