Featured Products
Our Promise to You
Guaranteed product quality, expert customer support

ONLINE INQUIRY
Immortalized Mouse Natural Killer Cells (KIL C.2)
Cat.No.: CSC-I9259L
Species: Mus musculus
Source: Blood
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
2) Wright-Giemsa stain to visualize cells;
3) Western blost used to detect granzyme B.
The Immortalized Mouse Natural Killer Cells (KIL C.2) are an immortalized cell line derived from CD3-NK1.1+ NK cells in mouse bone marrow, developed through co-culture with OP-9S/LMJSN and post-5-FU bone marrow MNCs. NK cells are crucial immune cells, classified as lymphocytes, typically originating from bone marrow and developing in the thymus or during embryogenesis. They are distributed across various tissues, including the bone marrow, liver, thymus, with smaller amounts found in the spleen, lymph nodes, and peripheral blood. These cells can directly kill tumor cells and virus-infected cells without antigen sensitization, playing a vital role in the body's innate immune defense.
KIL C.2 cells retain most characteristics of normal immature NK cells, including the ability to secrete perforin and granzymes, thereby inducing target cell lysis or apoptosis. In research, KIL C.2 serves as a model for studying mouse NK cells' roles in cancer and viral infections, including investigating NK cell recognition and killing mechanisms against tumor cells and developing NK cell-based tumor immunotherapy strategies. Additionally, it has been used to study the impact of MDSCs (myeloid-derived suppressor cells) on NK cell function and can be employed to study interactions between NK cells and other immune cells, such as MOC2 cells, via transfection or co-culture.
Kbtbd2 Inhibits Proliferation of NK Cell Line
HCC ranks as a significant cancer threat, with NK cell therapies being a potent treatment option. However, NK cell function is compromised in HCC environments due to immune suppressive mechanisms, with underlying molecular determinants not fully identified. Dai's team utilized a Tet-on inducible expression system in the NK cell line KIL C.2 to control Kbtbd2 expression. This allowed assessment of Kbtbd2's effects on proliferation, receptor expression, and cytotoxicity under controlled conditions, compared to non-expressing cells. The modified KIL C.2 cells in this system, termed KILC (without exogenous Kbtbd2 coding sequence) or KILK (with exogenous Kbtbd2 coding sequence), were used for the following tests. Without doxycycline treatment, KILC and KILK cells expressed very low Kbtbd2, while doxycycline treatment substantially induced high Kbtbd2 expression in KILK cells only (Fig. 1C). Kbtbd2 was present in both cytoplasm and nucleus (Fig. 1D). Doxycycline treatment induced no significant difference in apoptosis of either KILC or KILK cells (Fig. 1E). The proliferation of this cell line was then analyzed using Ki67 staining after stimulation with 20 ng/ml IL-2 in the presence or absence of doxycycline for 48 hours. IL-2 elicited more than 50% of proliferative cells (Fig. 1F). Doxycycline treatment slowed down IL-2-induced KILK cell proliferation (Fig. 1F).
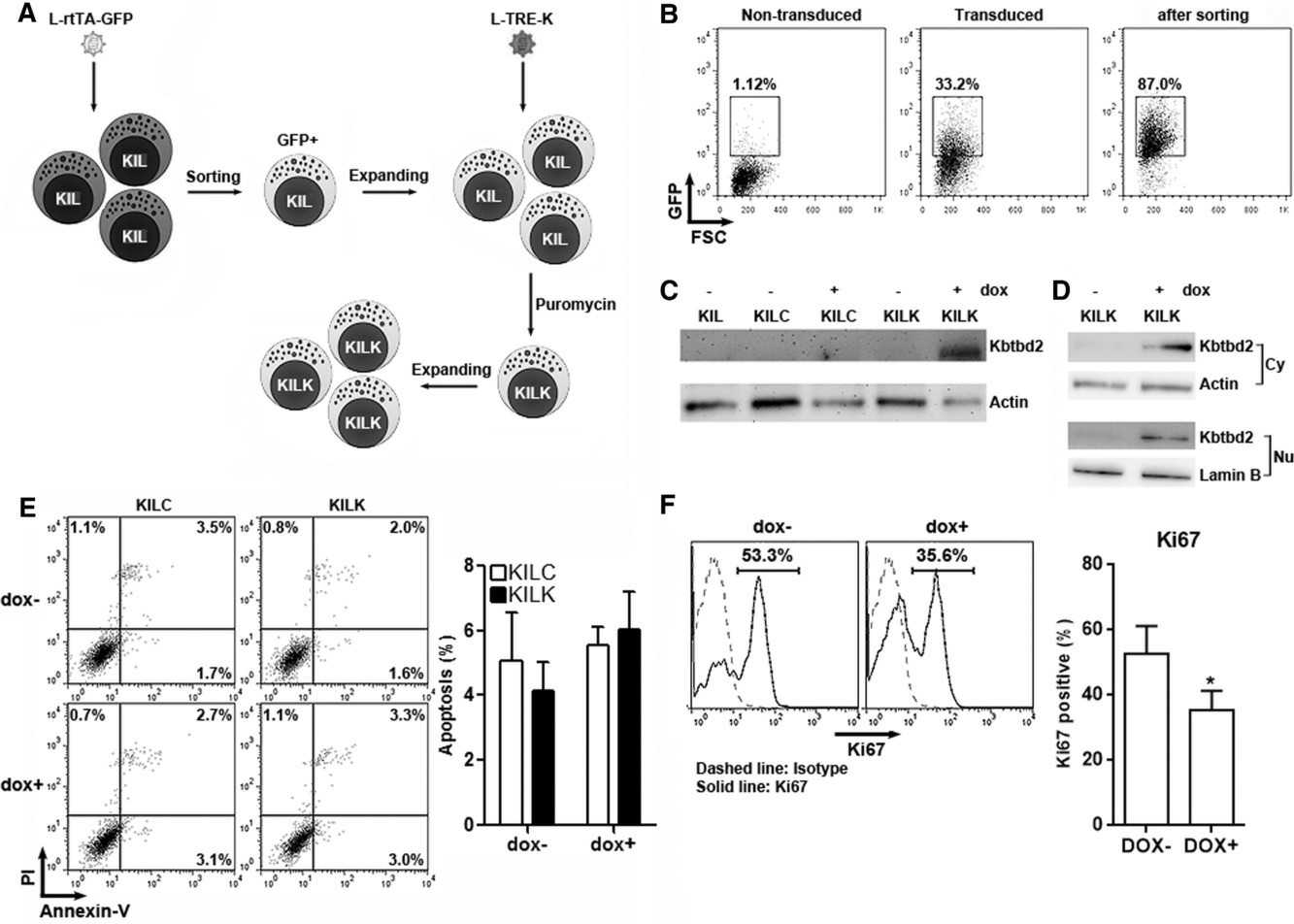 Fig. 1. Effect of exogenous Kbtbd2 on NK cell line proliferation (Dai K, Huang Y, et al., 2018).
Fig. 1. Effect of exogenous Kbtbd2 on NK cell line proliferation (Dai K, Huang Y, et al., 2018).
Establishment and Characterization of KIL C.1-C.5
In co-cultures of post-5-FU bone marrow MNCs with OP-9S/LMJSN, CD3-NK1.1+ NK cells showed significant proliferative capacity, becoming predominant within 2-3 months, unlike the CD19+B220+ B lymphoid cells in control OP-9S/LXSN. From the OP-9S/LMJSN cultures, spontaneously immortalized NK precursor lines, such as KIL (killer lymphocyte), emerged. KIL has been maintained for 2 years, exhibiting a racket shape at 37°C (Fig. 2A). Dependency on IL-7 for survival and proliferation was noted, with rapid apoptosis without it (Fig. 2B). IL-7 combined with SCF enhanced proliferation (Fig. 2B-C). KIL expresses CD45, CD25, NK1.1, but not CD3, CD4, CD8, CD19 (Fig. 3A). The TCRαβ loci remain in germline configuration (Fig. 3B-C). Five clonal lines of KIL, designated KIL C.1-C.5, were obtained by limiting dilution. KIL C.2 was chosen as the prototype because it had a shorter doubling time. KIL C.2 is very similar to the un-cloned KIL in every respect examined, including morphology, growth factor responsiveness, cell surface markers, and cytotoxicity. In addition to all surface markers shown in Figure 3A, both KIL C.2 and the parental KIL also express CD43, CD51, and CD94 (Fig. 4) and NKG2D.
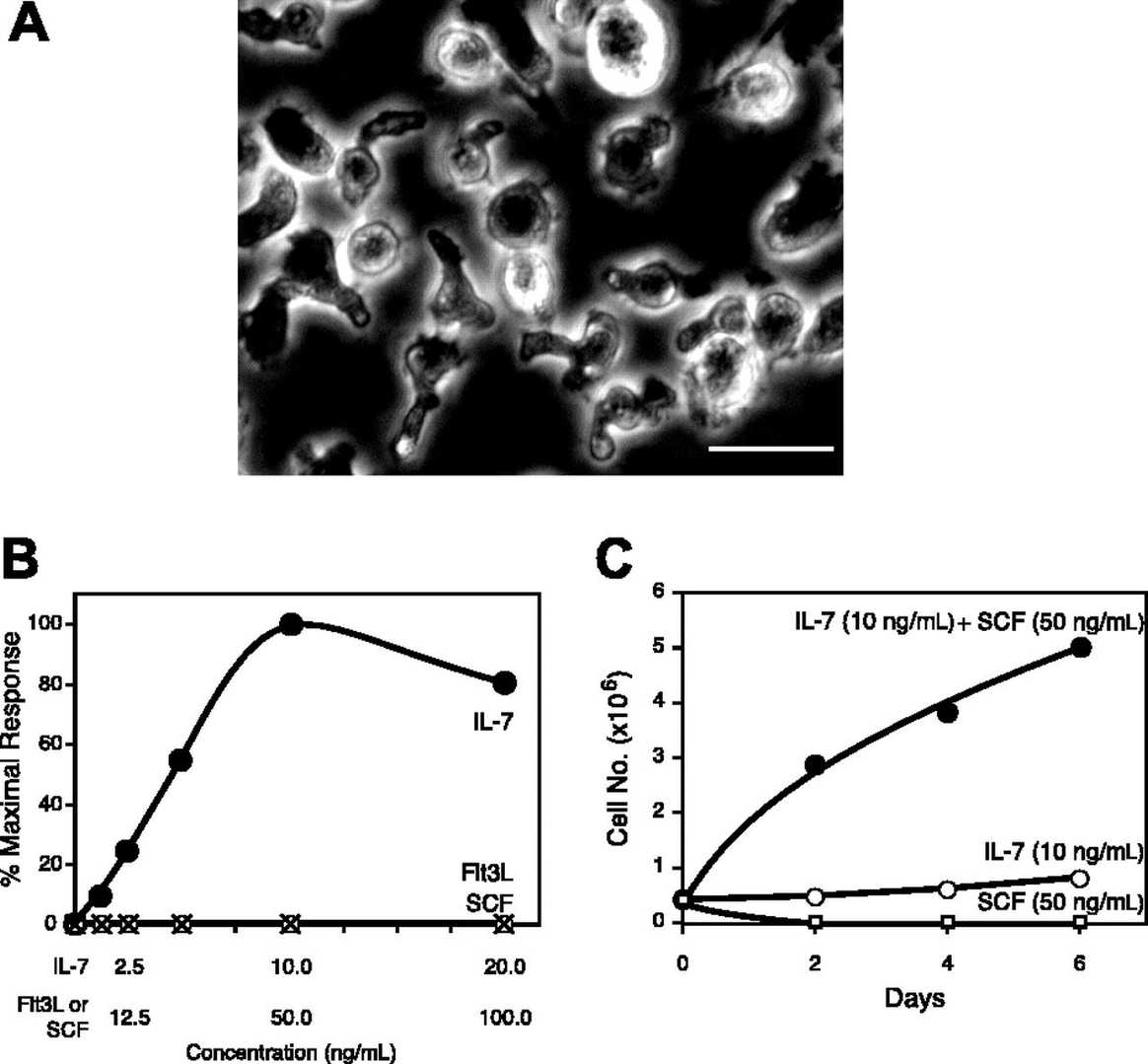 Fig. 2. Morphology and growth factor responses of KIL (DeHart S L, Heikens M J, et al., 2005).
Fig. 2. Morphology and growth factor responses of KIL (DeHart S L, Heikens M J, et al., 2005).
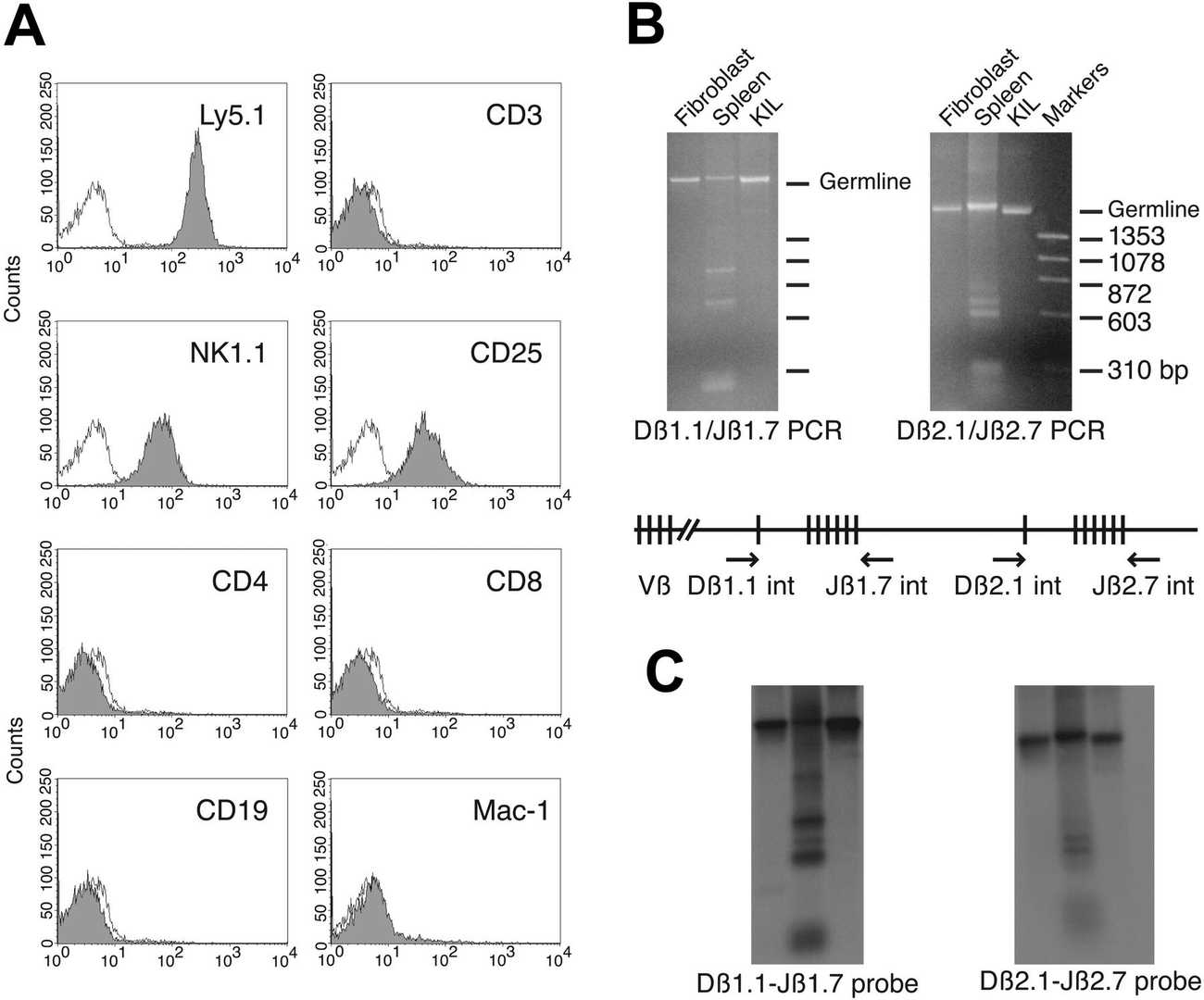 Fig. 3. Phenotypic markers of the KIL cell line (DeHart S L, Heikens M J, et al., 2005).
Fig. 3. Phenotypic markers of the KIL cell line (DeHart S L, Heikens M J, et al., 2005).
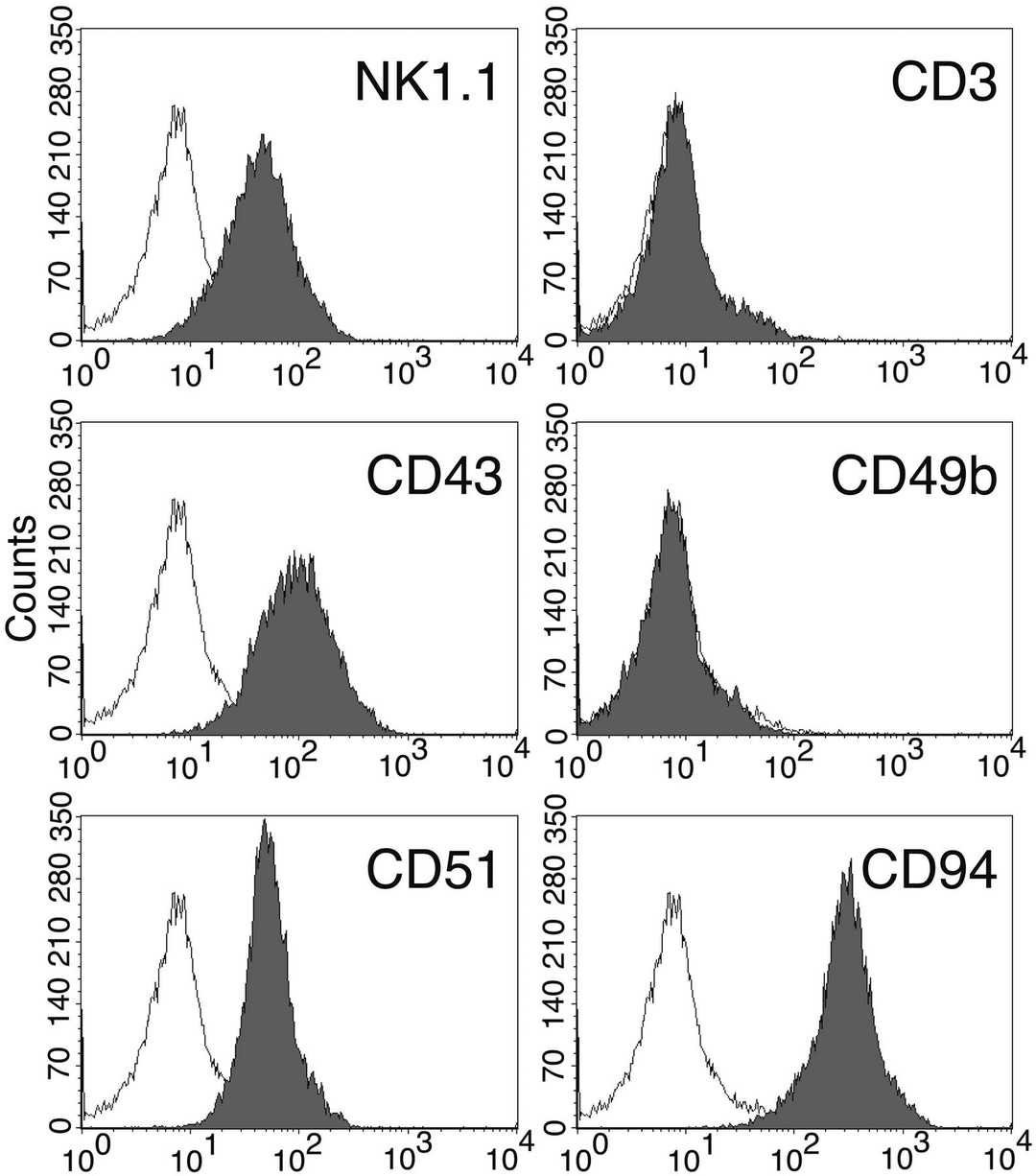 Fig. 4. Cell surface markers of the KIL C.2 clone (DeHart S L, Heikens M J, et al., 2005).
Fig. 4. Cell surface markers of the KIL C.2 clone (DeHart S L, Heikens M J, et al., 2005).
Ask a Question
Write your own review

